|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Monographs High School High School Chemistry Chemistry Matter Matter  |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Created: 30 Jul, 2010; Last Modified: 10 Aug, 2021 The page has been upgraded to a new home. Please follow this link. Matter - 05Properties of GasesBecause the distance between the particles of a gas are orders of magnitude more than the distance between particles in liquids and solids, gases have certain unique properties. These are as follows:
The Kinetic-Molecular Theory of Gas
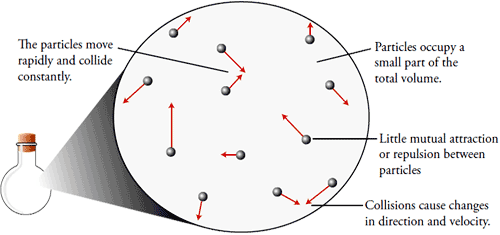
The properties of gases as outlined above have their explanations based on the kinetic-molecular theory of gas (KMT), which is essentially the kinetic theory of matter applied to gases (see Fig. ). According to this theory, the particles of a gas are in constant rapid and random motion. They move in straight lines, till they collide with each other or with the walls of the container, which changes their direction of motion. The particles are very far apart relative to their sizes. This explains the fluidity, low density and compressibility of gases. This theory states that the pressure exerted by a gas is due to the frequency of collisions of the gas particles with each other and the walls of its container. Gas PressureOur atmosphere, or air, is a mixture of gases, and envelopes the earth. The air particles are pulled towards the earth due to gravity, making the air denser at lower altitudes than at higher altitudes. The collisions of the air particles with anything in contact with the atmosphere, such as the earth's surface, results in atmospheric pressure or air pressure. Since the atmosphere is denser at lower altitudes, the atmospheric pressure at sea level, for example, will be higher than at the top of a mountain. Measuring pressurePressure is defined as the force per unit area. In SI units, the unit of force is the newton (N), which is the force that gives an object of mass The SI unit of pressure is the pascal (Pa), which is the the force of Pressure is measured using an instrument called a barometer. Mercury Barometer
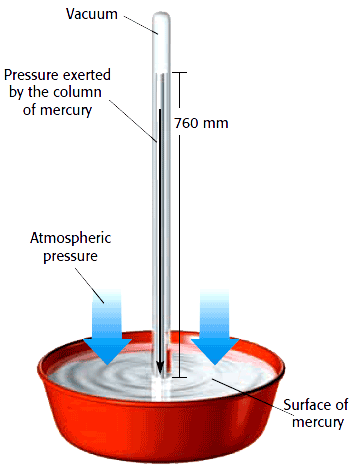
A mercury barometer consists of a long tube closed at one end, filled with mercury (Hg), and inverted in a vessel containing mercury (see Fig. ). As depicted in the figure, the air exerts pressure on the surface of mercury in the vessel. When the tube filled with mercury is inverted into the pool of mercury (taking care not to allow air in), the mercury in the tube falls till the pressure exerted downwards by the weight of the mercury column balances the atmospheric pressure at the surface of the pool. At sea level, it is found that the air pressure can support a mercury column height of This pressure is read as
.......(1)
STP – standard temperature and pressureFor studying changes in properties of gases with changing temperatures and pressures, a standard for comparison purposes is useful. The scientific community uses a set of standard conditions called standard temperature and pressure, or STP, which stands for a temperature of The Gas LawsUnlike solids and liquids, gases exhibit remarkably similar behaviour regardless of their chemical structure. In the 17th century, scientists found out that the physical properties of gases can be defined using four variables: pressure (P), volume (V), temperature (T) and amount, or number of moles (n). The gas laws define the relationships between these variables. A gas which exactly follows these relationships is called an ideal gas. There are four main gas laws – Boyle's law, Charles's law, Gay-Lussac's law and Avogadro's law. The last two laws will be dealt with later on in the subject. Boyle's Law: pressure-volume relationship
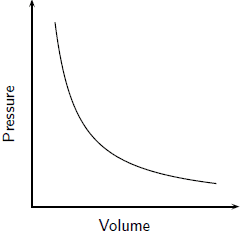
In 1662, the English scientist Robert Boyle found that, for a gas in a container maintained at a constant temperature, the volume of the gas decreases with rising pressure. The pressure-volume relationship is an example of an inverse relationship, in which increasing the value of one parameter decreases that of the other, and vice-versa. (see Fig. ) On the basis of the KMT, decreasing the volume of a gas implies that there will be more frequent collisions on the walls of the container by the gas particles since the number of particles is remaining constant. Hence, the pressure, which is a measure of the frequency collisions of the gas particles with the walls of the container, goes up. The opposite is true when the volume of the gas is increased.
Boyle, on taking accurate measurements, further found that not only are pressure and volume inversely related, but at constant temperature, they are inversely proportional. A graph of the pressure against the inverse of volume came out to be a straight line (Fig. ).
Boyle's law:
At constant temperature, the pressure of an ideal gas is inversely proportional to its volume.
Thus,
where: P = pressure, V = volume, k = proportionality constant Rearranging, we get the mathematical formulation for Boyle's law:
.......(2)
Thus, according to Boyle's law, for ideal gases at constant temperature, the product of pressure and volume is constant. A useful form of the above equation, for the purpose of solving problems, is:
.......(3)
where: P
A sample of helium occupies a volume of We have:
Boyle's law is applicable since this is a constant temperature process. Thus,
Rearranging to make V
Substituting the values:
Thus, the final volume occupied by helium is Note 1: In examples like this one, it was not necessary to convert units to SI units during the calculation process. This is so because the change of pressures or volumes to SI units require multiplication with conversion factors, which can as easily be applied to the final result, if the result is required to be in SI units. However, you should make sure that the units should be used consistently throughout the calculation process. Note 2: The rules for significant figures arithmetic have not been applied. Charles's Law: volume-temperature relationship
Around 1787, Jacques Charles had found that, at constant pressure, the volume of a gas increases with rising temperature. This volume-temperature relationship is an example of a direct relationship, in which increasing the value of one parameter increases that of the other. On the basis of the KMT, increasing the temperature of a gas results in the gas particles becoming more energetic, which increases the frequency and the force of collisions on the walls of the container by the gas particles. If the container walls are flexible, the container will tend to increase in volume. Charles found that not only are volume and temperature directly related, but at constant pressure, volume and absolute temperature are directly proportional. A graph of the volume against temperature is a straight line (see Fig. ). From the graph it might appear that as the absolute temperature reduces to Charles's law:
At constant pressure, the volume of an ideal gas is directly proportional to its absolute temperature.
Thus, where: V = volume, T = temperature, k = proportionality constant Rearranging, we get the mathematical formulation for Charles's law:
.......(4)
Thus, according to Charles's law, for ideal gases at constant pressure, the division of volume by temperature is constant. A useful form of the above equation, for the purpose of solving problems, is:
.......(5)
where: V
At a temperature of We have:
Charles's law is applicable since this is a constant pressure process. Thus,
Rearranging to make V
Substituting the values:
Thus, the final volume occupied by carbon dioxide is Note 1: In this example, the conversion of temperature from celsius to the SI unit of kelvins is required, because the conversion for temperature requires addition of a fixed conversion term, not multiplication by a fixed factor, as in the case of pressure and volume. Note 2: The rules for significant figures arithmetic have not been applied. The General Gas Law EquationBoyle's Law relates the pressure and volume of a gas at constant temperature,
.......(6)
A useful form of the above equation is:
.......(7)
where: P
At the beginning of a journey, a truck tyre has a volume of We have:
The general gas law equation can be used to solve this problem. Thus,
Rearranging to make P
Substituting the values:
Thus, the final pressure of the gas is Note: The rules for significant figures arithmetic have not been applied. Feedback
If your email client is configured, click here to send your feedback.
Otherwise, send an email to feedback@mentorials.com with subject line: "Feedback: Properties of Gases & Gas Laws". List of ReferencesBishop, M, An Introduction to Chemistry (First Version), viewed 10 February 2007 <http://preparatorychemistry.com/Bishop_Chemistry_First.htm>, 2005. FHSST, Chemistry Grades 10 - 12, ver 0, viewed 10 March, 2009, <http://www.fhsst.org>, 2008. Myers, RT, Oldham, KB & Tocci, S, Holt Chemistry, USA:Holt, Rinehart and Winston, 2006. BibliographyBishop, M, An Introduction to Chemistry (First Version), viewed 10 February 2007 <http://preparatorychemistry.com/Bishop_Chemistry_First.htm>, 2005. FHSST, Chemistry Grades 10 - 12, ver 0, viewed 10 March, 2009, <http://www.fhsst.org>, 2008. McMurray, J & Fay, RC, Chemistry, 4th edn, USA: Prentice Hall, 2003. Myers, RT, Oldham, KB & Tocci, S, Holt Chemistry, USA:Holt, Rinehart and Winston, 2006. Nathan, HD & Henrickson, C, CliffsQuickReview Chemistry, NY, USA: Hungry Minds, 2001. Uvarov, EB & Chapman, DR, A Dictionary of Science, 5th edn, UK: The Chauser Press, 1979. Whitten, KW, Davis, RE, Peck, L & Stanley, GG, General Chemistry, 7th edn, Belmont, USA: Thomson Brooks/Cole, 2004.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Monographs High School High School Chemistry Chemistry Matter Matter  |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Copyright © 2009-2025 Mentorials.com. All Rights Reserved.
[Site best viewed at 1024 x 768 resolution, using Mozilla Firefox 3.5 and above] Some clipart images used herein were obtained from IMSI's MasteClips® and MasterPhotos™ Premium Image Collection. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||

 High School
High School Mathematics
Mathematics