|
|||||||||||||||||||
|
|||||||||||||||||||
Monographs High School High School Chemistry Chemistry Matter Matter  |
|||||||||||||||||||
Created: 20 Jan, 2010; Last Modified: 10 Aug, 2021 The page has been upgraded to a new home. Please follow this link. Matter - 02Separation of MixturesIn a mixture, since the constituent substances do not lose their identity, they can be separated easily by physical methods, taking advantage of the differences in their physical properties.
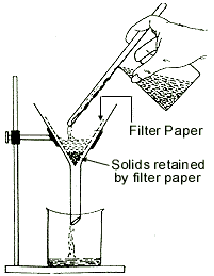
Mixture of Solids and LiquidsSedimentation and DecantationSedimentation is the process of separating an insoluble solid from a liquid in which it is suspended by allowing it to settle to the bottom of the container. If this also involves pouring off of the liquid leaving the solid behind, it is called decantation. FiltrationFiltration is used for separating insoluble solids from a liquid. When a mixture of chalk and water is poured through a filter paper in a funnel, chalk particles remains as residue in the filter paper, while the water gets collected in the beaker below as filtrate. (see Fig. ) EvaporationEvaporation is used for recovering dissolved solid substances from solutions by evaporating the solvent. The solute “dissolves out” and is left behind. Sugar can be recovered from a sugar-water mixture by evaporation. The water evaporates to leave the solute behind. Copper sulphate, potash alum, potassium nitrate etc. can also be recovered from their aqueous solutions by evaporation.
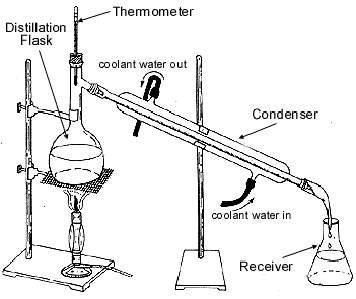
CrystallisationCrystallisation is a sophisticated form of evaporation technique in which crystals of the solute are encouraged to develop during the process of “dissolving out” from the solution as the solvent evaporates. DistillationDistillation is the process of heating a solution containing soluble solids to form vapours of the liquid and then cooling the vapours to get the liquid back. A mixture of common salt and water is taken in a distillation flask and heated. Steam rises up and comes out into the condenser, which is a coaxial tube with a central tube for vapour to pass through, and an outer tube through which cold water circulates to form a cold water jacket. Steam condenses in the central tube and collects in a receiver as distillate. (see Fig. )
Mixture of SolidsSublimationSublimation is a process in which some solids, on heating, are transformed directly to vapour without passing through the liquid phase. This technique can be used to separate a mixture of solids, one of which can undergo sublimation. The vapours are then cooled separately to get the sublimed solid back, a process called deposition. (Fig. ) Sublimation is used in the separation of substances like ammonium chloride, iodine, napthalene, camphor and sulphur. ExtractionIn some cases, one substance in the mixture is soluble in a particular liquid solvent whereas the other(s) is(are) insoluble in it. The process of dissolving out the soluble component from a mixture, and subsequently treating the solution to get the solid, is called extraction. Magnetic SeparationThis method of separation is exemplified by the separation of iron filings. A mixture with iron filings as one of the components can be separated using a magnet to attract the iron particles away from the mixture.
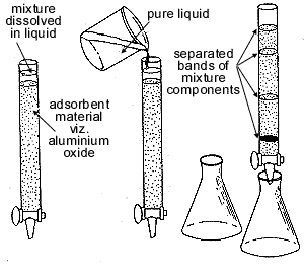
ChromatographyChromatography is an advanced technique of separation in which individual components of a mixture are separated from each other using the property of differential migration (different rates of flow). Here, a mobile phase, carrying the mixture, is passed through a selectively adsorbentAn adsorbent substance has the capability of retaining particles of other solids, liquids or gases on its surface, as opposed to the process of absorption in which the particles are taken inside the bulk of the absorbing substance. stationary phase, which can retain the components of the mixture to different degrees. (Fig. ) Mixture of LiquidsFractional Distillation
Fractional distillation is the process of separating two or more miscible liquids by a modified distillation process, in which the distillates are collected as fractions having different boiling points. The separation of the liquids by this method is based on the difference in their boiling points. (Fig. ) Fractional distillation makes use of a fractionating column or distillation column, a tube which provides different temperature zones inside it during distillation, the temperature decreasing from bottom to top. It provides surfaces on which condensations (of less volatileA volatile substance is one which tends to easily evaporate liquids) and vaporizations (of more volatile liquids) can occur before the vapours enter the condenser in order to concentrate the more volatile liquid in the first fractions and the less volatile components in the later fractions. Fractional distillation is very effective is separating mixtures of volatile components, and is widely used in laboratories and industries. Crude petroleum is separated by the process of fractional distillation into different fractions like gasoline, lubricating oil, kerosene, diesel etc.
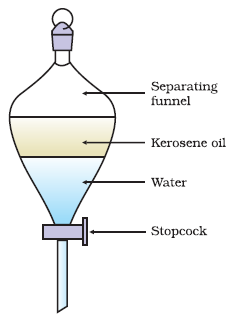
Gravity SeparationIn gravity separation, a mixture of two immiscible liquids can be separated using a separating funnel, the working of which is based on the differences in the densities of the liquids. The heavier liquid which settles below is drained out first from below the funnel into a beaker, and then the lighter liquid is drained out into another beaker.
Feedback
If your email client is configured, click here to send your feedback.
Otherwise, send an email to feedback@mentorials.com with subject line: "Feedback: Separation of Mixtures". List of ReferencesNCERT, Science – Textbook for Class IX, viewed 25 February, 2008, <http://www.ncert.nic.in/textbooks/testing/Index.htm>, (n.d.). BibliographyNCERT, Science – Textbook for Class IX, viewed 25 February, 2008, <http://www.ncert.nic.in/textbooks/testing/Index.htm>, (n.d.).
|
|||||||||||||||||||
Monographs High School High School Chemistry Chemistry Matter Matter  |
|||||||||||||||||||
|
Copyright © 2009-2025 Mentorials.com. All Rights Reserved.
[Site best viewed at 1024 x 768 resolution, using Mozilla Firefox 3.5 and above] Some clipart images used herein were obtained from IMSI's MasteClips® and MasterPhotos™ Premium Image Collection. |
|||||||||||||||||||

 High School
High School Mathematics
Mathematics